
At 64 years of age, I returned to university with the intention of developing an understanding of the physiology of the joint disorders that accompany Lyme disease.
This search led me to an honours degree in Biology with a minor in Chemistry. My fourth year thesis investigated the ability of Cinchona alkaloids and two laboratory produced analogues to block potassium channels in neurons. In order to visualize this blocking process in vivo, I developed a technique for driving fluorescent labeled potassium ions through neural tissue using the neuron's electrical potential in the neural ganglia of leech Nephalopsis obscura . The leeches were exposed to the chemical for a period of 1 minute, then anaesthatized (TricaineMethylSulphonate in 10% ethanol) and the ventral nerve cord was dissected out. Lucifer yellow potasium stain was introduced through a pulled glass siphon attached to a steel anode and place at the anterior portion of the ganglion. A copper cathode was set at the posterior portion of the ganglion.
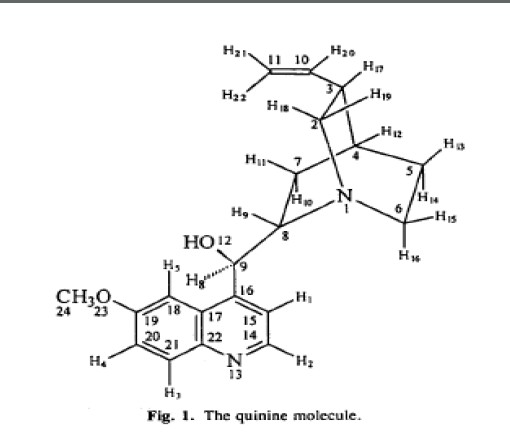
 This a central ganglion from an untreated leech .
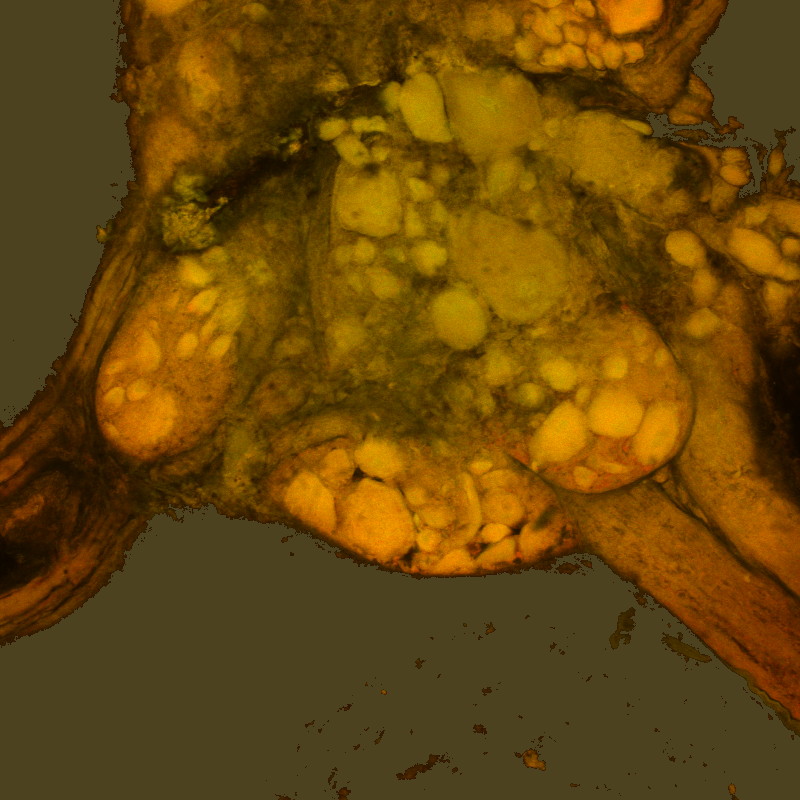
This a central ganglion from an untreated leech . 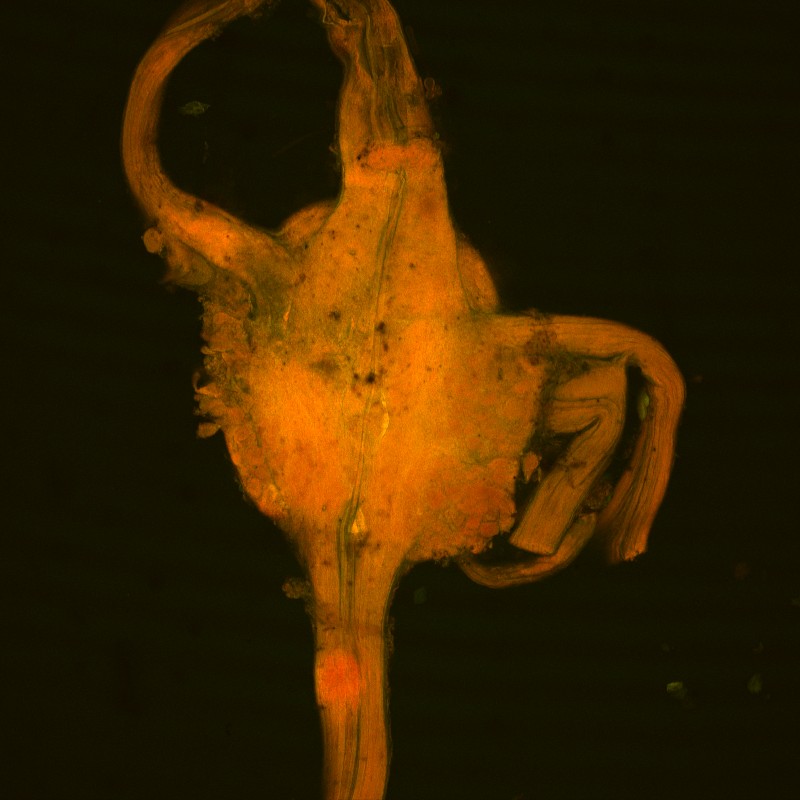 Ganglion from a leech that was exposed to (+)-cinchonine for 1minute(Neural sheath intact).
Ganglion from a leech that was exposed to (+)-cinchonine for 1minute(Neural sheath intact).

 Ganglia from a leech that was exposed to methylated (+)-cinchonine for 1minute.
Ganglia from a leech that was exposed to methylated (+)-cinchonine for 1minute.
In completing the degree requirements, I continued the investigation of Cinchona alkaloids with a photosensitivity study of -(-)quinine. In this research I established through NMR analysis that the quinoline and quinuclidine structures remain intact and joined by a single carbon so that the superoxide radical produced through UV exposure comprises the release of the hydroxyl group on the central carbon.
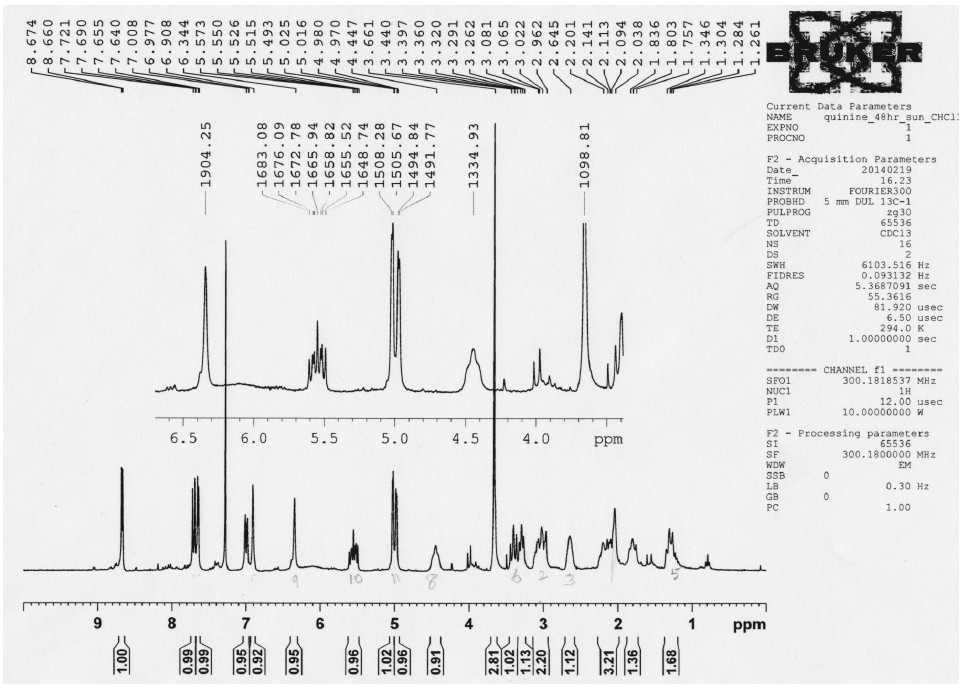
Figure 11. This Proton NMR is as that described in Figure 9 with an increase length of exposure time to 48 hours under sunlight. The shift noted above has increased with the proton on carbon 8 at δ 4.447 and that on carbon 9 at δ 6.344. The protons of the quinoline substructure have also shifted slightly the proton at carbon 14 remains the same at δ 8,674 and that at carbon 21 also remains the same at δ 7.721 but the proton at carbon 15 has shifted downfield to δ 7.655 from δ 7.545. The remaining 2 protons on either side of the methoxy position appear to have move slightly upfield.
These results suggested that the methylation of this hydroxyl group would stabilize the quinine structure and interfere with the mechanism for its release of a superoxide anion. It is my opinion that beta blockers such as quinine and propranolol are important meuromodulators that may affect bone remodeleng in respose to hypothalmus activation.
Home Neural Research Lyme Disease Quinine Chaga Mushroom Contact
Jane's Pottery